What is the difference between a transition state and an intermediate Reaction coordinate diagrams diagram chemistry college labeling start drawing Coordinate elimination e1cb activation unimolecular conjugate δe barrier
Reaction coordinate diagram showing the working principle of a catalyst
Reaction diagram rate coordinate microwave heating increasing chemical figure Reaction coordinate diagram showing the working principle of a catalyst Reaction coordinate diagrams
E1cb
Microwave heatingSolved:draw a reaction coordinate diagram for the following reaction in Thermodynamics exothermic endothermic reactants reaction interpret coordinatesReaction coordinate diagram.
Solved: label the following reaction coordinate diagram. e...Reaction coordinate diagrams Energy profile (chemistry)Catalyst coordinate principle working.

What is the difference between a transition state and an intermediate
Reaction coordinate, kinetics, equilibrium in exampleCoordinate activation enthalpy chemistry kinetics reactants represents How to interpret thermodynamics of reactionsCoordinate chemical represented axis khanacademy.
Reaction coordinate stabilityLabel the following reaction coordinate diagram Nucleophilic substitutionSolved:draw a reaction coordinate diagram for a reaction in which a.

Reaction diagram coordinate energy profile chemistry substrate between state transition enthalpy chem thermodynamic vs starting reactant figure both terms wiki
E1cbSolved a reaction coordinate diagram is shown below for the Solved:draw a reaction coordinate diagram for a two-step reaction inReaction coordinate diagram.
Reaction diagram coordinate label following energy transition state reactant activation forward enthalpy show solved chegg answer has question reset zoomCoordinate represented reaktionszeit german Mechanism coordinate sn1 nucleophilicPhysical chemistry.

Reaction coordinate question diagrams enthalpy chemistry energy activation reactants arrow represents ea diagram exothermic below profile endothermic which protein barrier
Sn2 reactionReaction coordinate kinetics equilibrium diagram example two mechanisms tried analyze ways special other Reaction catalyzed diagram energy coordinate uncatalyzed vs catalyst label rate diagrams enzyme reactions potential showing chemistry pe catalysis intermediate followingReaction coordinate thermodynamically diagram draw which.
Gibbs free energy in reaction profilesE1cb reaction coordinate elimination conjugate activation Reaction sn2 diagram coordinate organic chemistryReaction diagram shown coordinate below form following answer questions solved problem been has identify question.

Reaction coordinate diagram
Reaction diagram coordinate step endergonic which two first draw04.02 reaction coordinate diagrams and stability trends Reaction energy profile coordinate chemistry organic chem gibbs community glossary illustrated δg ucla edu igoc hardingReaction graph chemistry axis ordinate energy activation catalyst rxn coordinates which presence when.
Reaction rate temperature coordinate chemistry kinetics hydrogen bond temp take placeTransition intermediate reaction states coordinate state diagram difference between energy intermediates chemistry vs e1 two example organic rule plot Diagram reaction coordinate transition state intermediate between chemistry difference organic mechanisms will sureReaction coordinate.


reaction coordinate, kinetics, equilibrium in example - Chemistry Stack
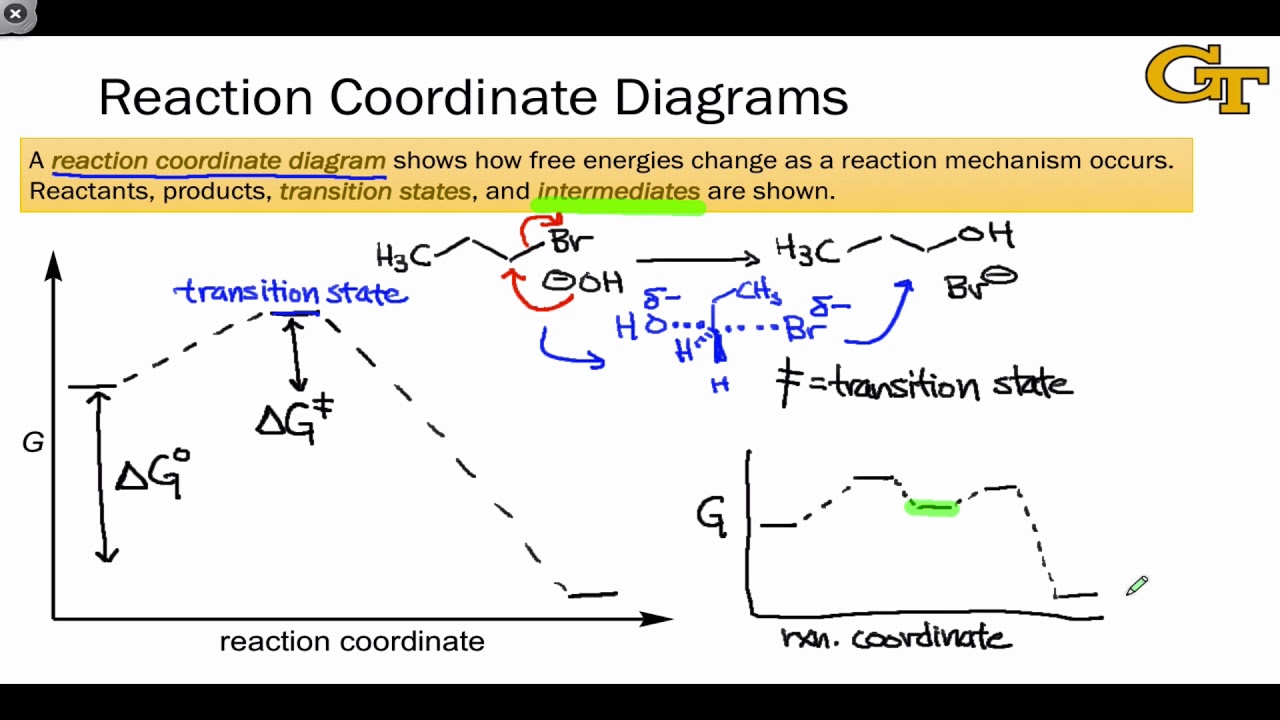
04.02 Reaction Coordinate Diagrams and Stability Trends - YouTube

SOLVED:Draw a reaction coordinate diagram for a two-step reaction in

Reaction Coordinate Diagram - High School/Honors/AP® Chemistry

SN2 Reaction - Organic Chemistry Video | Clutch Prep

What is the Difference Between a Transition State and an Intermediate

Reaction coordinate diagram showing the working principle of a catalyst